HistGen: Histopathology Report Generation via Local-Global Feature Encoding and Cross-modal Context Interaction
Mar 08, 2024Zhengrui Guo, Jiabo Ma, Yingxue Xu, Yihui Wang, Liansheng Wang, Hao Chen
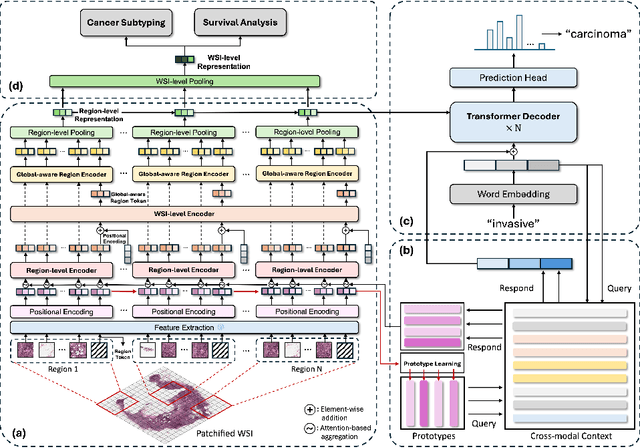
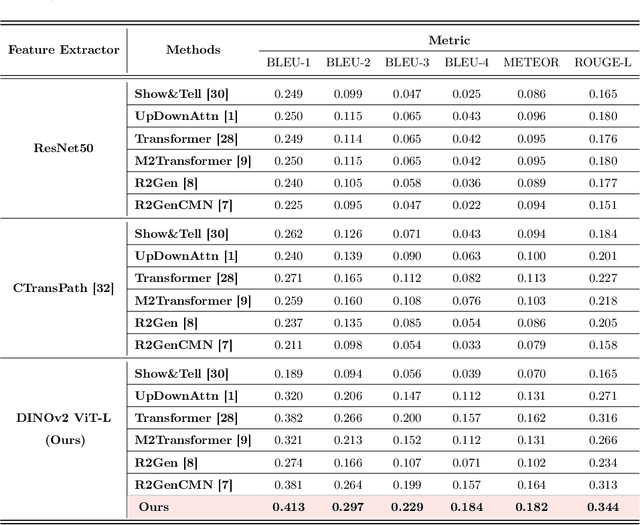
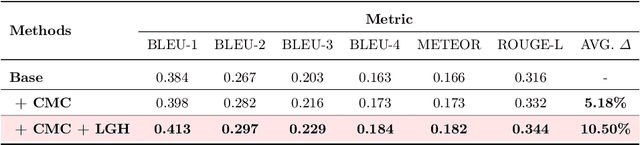
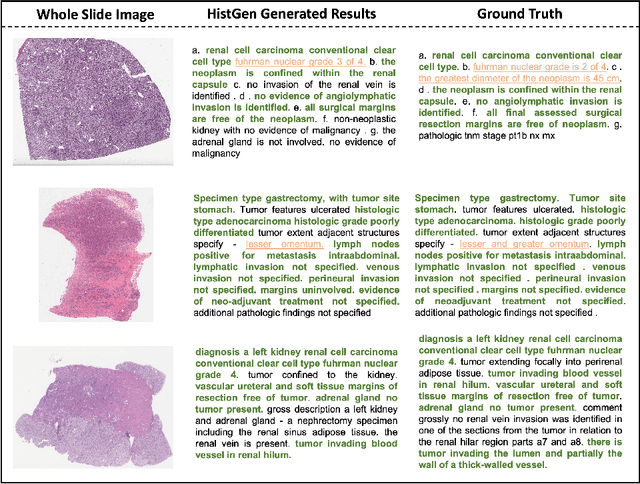
Histopathology serves as the gold standard in cancer diagnosis, with clinical reports being vital in interpreting and understanding this process, guiding cancer treatment and patient care. The automation of histopathology report generation with deep learning stands to significantly enhance clinical efficiency and lessen the labor-intensive, time-consuming burden on pathologists in report writing. In pursuit of this advancement, we introduce HistGen, a multiple instance learning-empowered framework for histopathology report generation together with the first benchmark dataset for evaluation. Inspired by diagnostic and report-writing workflows, HistGen features two delicately designed modules, aiming to boost report generation by aligning whole slide images (WSIs) and diagnostic reports from local and global granularity. To achieve this, a local-global hierarchical encoder is developed for efficient visual feature aggregation from a region-to-slide perspective. Meanwhile, a cross-modal context module is proposed to explicitly facilitate alignment and interaction between distinct modalities, effectively bridging the gap between the extensive visual sequences of WSIs and corresponding highly summarized reports. Experimental results on WSI report generation show the proposed model outperforms state-of-the-art (SOTA) models by a large margin. Moreover, the results of fine-tuning our model on cancer subtyping and survival analysis tasks further demonstrate superior performance compared to SOTA methods, showcasing strong transfer learning capability. Dataset, model weights, and source code are available in https://github.com/dddavid4real/HistGen.
LYSTO: The Lymphocyte Assessment Hackathon and Benchmark Dataset
Jan 16, 2023Yiping Jiao, Jeroen van der Laak, Shadi Albarqouni, Zhang Li, Tao Tan, Abhir Bhalerao, Jiabo Ma, Jiamei Sun, Johnathon Pocock, Josien P. W. Pluim, Navid Alemi Koohbanani, Raja Muhammad Saad Bashir, Shan E Ahmed Raza, Sibo Liu, Simon Graham, Suzanne Wetstein, Syed Ali Khurram, Thomas Watson, Nasir Rajpoot, Mitko Veta, Francesco Ciompi
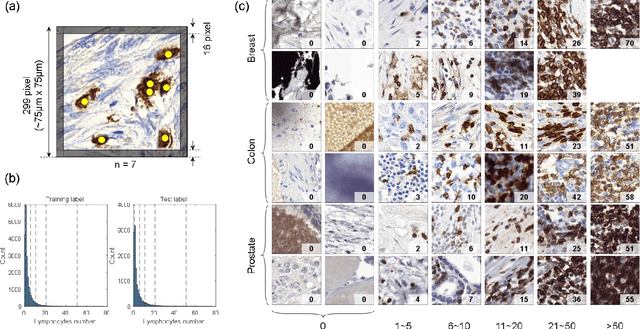
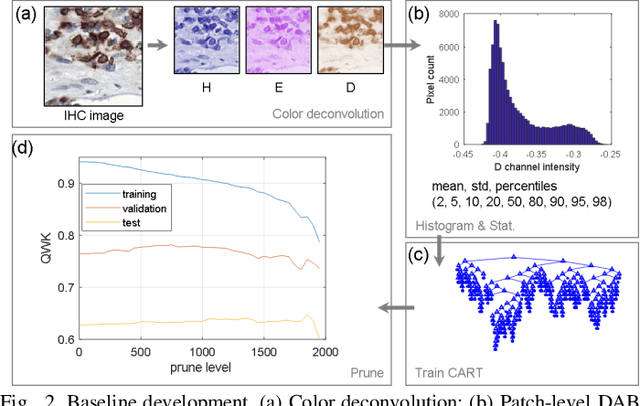
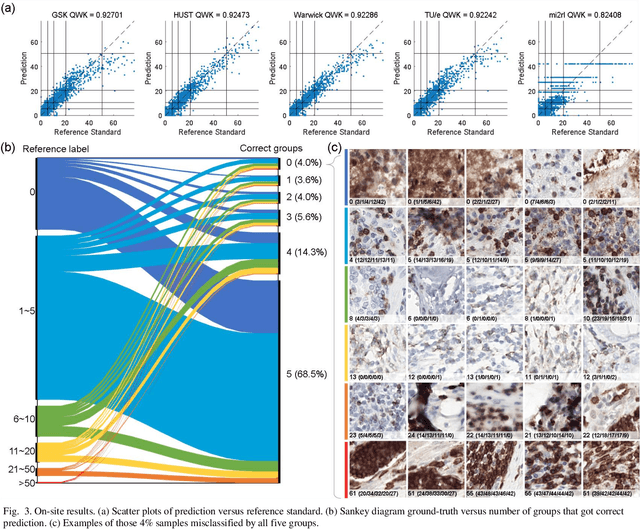
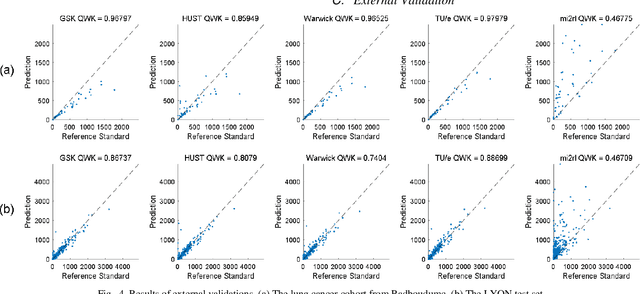
We introduce LYSTO, the Lymphocyte Assessment Hackathon, which was held in conjunction with the MICCAI 2019 Conference in Shenzen (China). The competition required participants to automatically assess the number of lymphocytes, in particular T-cells, in histopathological images of colon, breast, and prostate cancer stained with CD3 and CD8 immunohistochemistry. Differently from other challenges setup in medical image analysis, LYSTO participants were solely given a few hours to address this problem. In this paper, we describe the goal and the multi-phase organization of the hackathon; we describe the proposed methods and the on-site results. Additionally, we present post-competition results where we show how the presented methods perform on an independent set of lung cancer slides, which was not part of the initial competition, as well as a comparison on lymphocyte assessment between presented methods and a panel of pathologists. We show that some of the participants were capable to achieve pathologist-level performance at lymphocyte assessment. After the hackathon, LYSTO was left as a lightweight plug-and-play benchmark dataset on grand-challenge website, together with an automatic evaluation platform. LYSTO has supported a number of research in lymphocyte assessment in oncology. LYSTO will be a long-lasting educational challenge for deep learning and digital pathology, it is available at https://lysto.grand-challenge.org/.
Reconstruct high-resolution multi-focal plane images from a single 2D wide field image
Sep 21, 2020Jiabo Ma, Sibo Liu, Shenghua Cheng, Xiuli Liu, Li Cheng, Shaoqun Zeng




High-resolution 3D medical images are important for analysis and diagnosis, but axial scanning to acquire them is very time-consuming. In this paper, we propose a fast end-to-end multi-focal plane imaging network (MFPINet) to reconstruct high-resolution multi-focal plane images from a single 2D low-resolution wild filed image without relying on scanning. To acquire realistic MFP images fast, the proposed MFPINet adopts generative adversarial network framework and the strategies of post-sampling and refocusing all focal planes at one time. We conduct a series experiments on cytology microscopy images and demonstrate that MFPINet performs well on both axial refocusing and horizontal super resolution. Furthermore, MFPINet is approximately 24 times faster than current refocusing methods for reconstructing the same volume images. The proposed method has the potential to greatly increase the speed of high-resolution 3D imaging and expand the application of low-resolution wide-field images.
FFusionCGAN: An end-to-end fusion method for few-focus images using conditional GAN in cytopathological digital slides
Jan 03, 2020Xiebo Geng, Sibo Liua, Wei Han, Xu Li, Jiabo Ma, Jingya Yu, Xiuli Liu, Sahoqun Zeng, Li Chen, Shenghua Cheng




Multi-focus image fusion technologies compress different focus depth images into an image in which most objects are in focus. However, although existing image fusion techniques, including traditional algorithms and deep learning-based algorithms, can generate high-quality fused images, they need multiple images with different focus depths in the same field of view. This criterion may not be met in some cases where time efficiency is required or the hardware is insufficient. The problem is especially prominent in large-size whole slide images. This paper focused on the multi-focus image fusion of cytopathological digital slide images, and proposed a novel method for generating fused images from single-focus or few-focus images based on conditional generative adversarial network (GAN). Through the adversarial learning of the generator and discriminator, the method is capable of generating fused images with clear textures and large depth of field. Combined with the characteristics of cytopathological images, this paper designs a new generator architecture combining U-Net and DenseBlock, which can effectively improve the network's receptive field and comprehensively encode image features. Meanwhile, this paper develops a semantic segmentation network that identifies the blurred regions in cytopathological images. By integrating the network into the generative model, the quality of the generated fused images is effectively improved. Our method can generate fused images from only single-focus or few-focus images, thereby avoiding the problem of collecting multiple images of different focus depths with increased time and hardware costs. Furthermore, our model is designed to learn the direct mapping of input source images to fused images without the need to manually design complex activity level measurements and fusion rules as in traditional methods.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge