ECG-QA: A Comprehensive Question Answering Dataset Combined With Electrocardiogram
Jun 21, 2023Jungwoo Oh, Seongsu Bae, Gyubok Lee, Joon-myoung Kwon, Edward Choi




Question answering (QA) in the field of healthcare has received much attention due to significant advancements in natural language processing. However, existing healthcare QA datasets primarily focus on medical images, clinical notes, or structured electronic health record tables. This leaves the vast potential of combining electrocardiogram (ECG) data with these systems largely untapped. To address this gap, we present ECG-QA, the first QA dataset specifically designed for ECG analysis. The dataset comprises a total of 70 question templates that cover a wide range of clinically relevant ECG topics, each validated by an ECG expert to ensure their clinical utility. As a result, our dataset includes diverse ECG interpretation questions, including those that require a comparative analysis of two different ECGs. In addition, we have conducted numerous experiments to provide valuable insights for future research directions. We believe that ECG-QA will serve as a valuable resource for the development of intelligent QA systems capable of assisting clinicians in ECG interpretations.
Text-to-ECG: 12-Lead Electrocardiogram Synthesis conditioned on Clinical Text Reports
Mar 09, 2023Hyunseung Chung, Jiho Kim, Joon-myoung Kwon, Ki-Hyun Jeon, Min Sung Lee, Edward Choi




Electrocardiogram (ECG) synthesis is the area of research focused on generating realistic synthetic ECG signals for medical use without concerns over annotation costs or clinical data privacy restrictions. Traditional ECG generation models consider a single ECG lead and utilize GAN-based generative models. These models can only generate single lead samples and require separate training for each diagnosis class. The diagnosis classes of ECGs are insufficient to capture the intricate differences between ECGs depending on various features (e.g. patient demographic details, co-existing diagnosis classes, etc.). To alleviate these challenges, we present a text-to-ECG task, in which textual inputs are used to produce ECG outputs. Then we propose Auto-TTE, an autoregressive generative model conditioned on clinical text reports to synthesize 12-lead ECGs, for the first time to our knowledge. We compare the performance of our model with other representative models in text-to-speech and text-to-image. Experimental results show the superiority of our model in various quantitative evaluations and qualitative analysis. Finally, we conduct a user study with three board-certified cardiologists to confirm the fidelity and semantic alignment of generated samples. our code will be available at https://github.com/TClife/text_to_ecg
Automatic Detection of Noisy Electrocardiogram Signals without Explicit Noise Labels
Aug 08, 2022Radhika Dua, Jiyoung Lee, Joon-myoung Kwon, Edward Choi
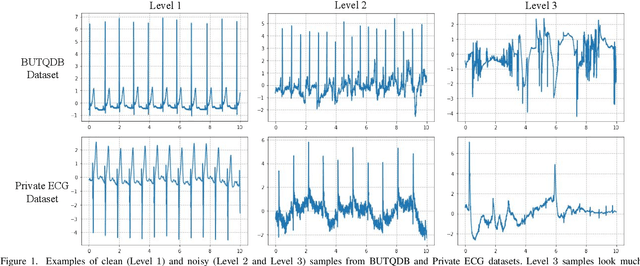
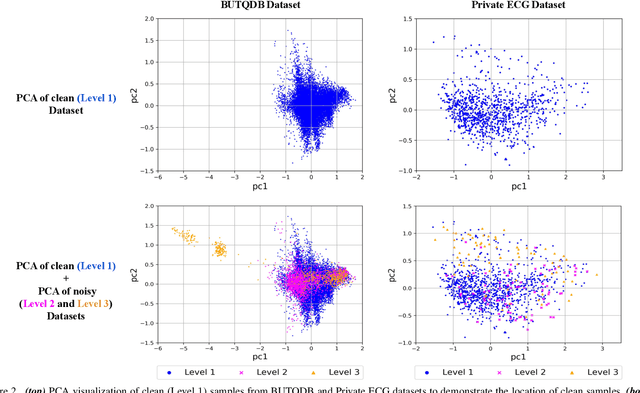
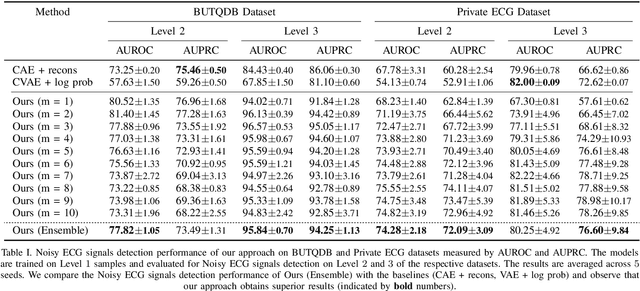
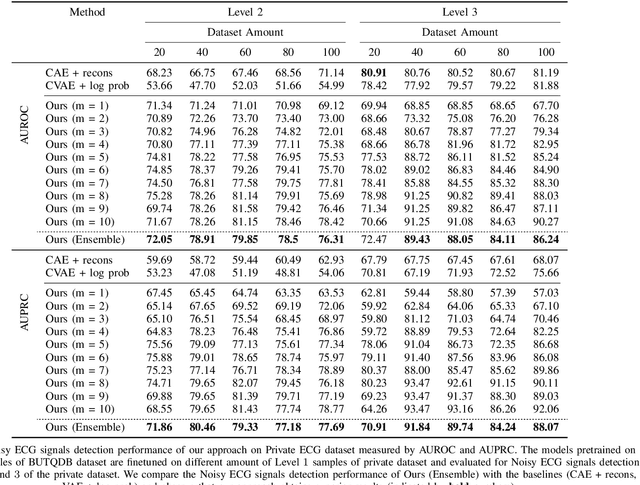
Electrocardiogram (ECG) signals are beneficial in diagnosing cardiovascular diseases, which are one of the leading causes of death. However, they are often contaminated by noise artifacts and affect the automatic and manual diagnosis process. Automatic deep learning-based examination of ECG signals can lead to inaccurate diagnosis, and manual analysis involves rejection of noisy ECG samples by clinicians, which might cost extra time. To address this limitation, we present a two-stage deep learning-based framework to automatically detect the noisy ECG samples. Through extensive experiments and analysis on two different datasets, we observe that the deep learning-based framework can detect slightly and highly noisy ECG samples effectively. We also study the transfer of the model learned on one dataset to another dataset and observe that the framework effectively detects noisy ECG samples.
Lead-agnostic Self-supervised Learning for Local and Global Representations of Electrocardiogram
Mar 18, 2022Jungwoo Oh, Hyunseung Chung, Joon-myoung Kwon, Dong-gyun Hong, Edward Choi




In recent years, self-supervised learning methods have shown significant improvement for pre-training with unlabeled data and have proven helpful for electrocardiogram signals. However, most previous pre-training methods for electrocardiogram focused on capturing only global contextual representations. This inhibits the models from learning fruitful representation of electrocardiogram, which results in poor performance on downstream tasks. Additionally, they cannot fine-tune the model with an arbitrary set of electrocardiogram leads unless the models were pre-trained on the same set of leads. In this work, we propose an ECG pre-training method that learns both local and global contextual representations for better generalizability and performance on downstream tasks. In addition, we propose random lead masking as an ECG-specific augmentation method to make our proposed model robust to an arbitrary set of leads. Experimental results on two downstream tasks, cardiac arrhythmia classification and patient identification, show that our proposed approach outperforms other state-of-the-art methods.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge