Multimodal Learning To Improve Cardiac Late Mechanical Activation Detection From Cine MR Images
Feb 28, 2024Jiarui Xing, Nian Wu, Kenneth Bilchick, Frederick Epstein, Miaomiao Zhang
This paper presents a multimodal deep learning framework that utilizes advanced image techniques to improve the performance of clinical analysis heavily dependent on routinely acquired standard images. More specifically, we develop a joint learning network that for the first time leverages the accuracy and reproducibility of myocardial strains obtained from Displacement Encoding with Stimulated Echo (DENSE) to guide the analysis of cine cardiac magnetic resonance (CMR) imaging in late mechanical activation (LMA) detection. An image registration network is utilized to acquire the knowledge of cardiac motions, an important feature estimator of strain values, from standard cine CMRs. Our framework consists of two major components: (i) a DENSE-supervised strain network leveraging latent motion features learned from a registration network to predict myocardial strains; and (ii) a LMA network taking advantage of the predicted strain for effective LMA detection. Experimental results show that our proposed work substantially improves the performance of strain analysis and LMA detection from cine CMR images, aligning more closely with the achievements of DENSE.
NeurEPDiff: Neural Operators to Predict Geodesics in Deformation Spaces
Mar 13, 2023Nian Wu, Miaomiao Zhang




This paper presents NeurEPDiff, a novel network to fast predict the geodesics in deformation spaces generated by a well known Euler-Poincar\'e differential equation (EPDiff). To achieve this, we develop a neural operator that for the first time learns the evolving trajectory of geodesic deformations parameterized in the tangent space of diffeomorphisms(a.k.a velocity fields). In contrast to previous methods that purely fit the training images, our proposed NeurEPDiff learns a nonlinear mapping function between the time-dependent velocity fields. A composition of integral operators and smooth activation functions is formulated in each layer of NeurEPDiff to effectively approximate such mappings. The fact that NeurEPDiff is able to rapidly provide the numerical solution of EPDiff (given any initial condition) results in a significantly reduced computational cost of geodesic shooting of diffeomorphisms in a high-dimensional image space. Additionally, the properties of discretiztion/resolution-invariant of NeurEPDiff make its performance generalizable to multiple image resolutions after being trained offline. We demonstrate the effectiveness of NeurEPDiff in registering two image datasets: 2D synthetic data and 3D brain resonance imaging (MRI). The registration accuracy and computational efficiency are compared with the state-of-the-art diffeomophic registration algorithms with geodesic shooting.
Predicting the protein-ligand affinity from molecular dynamics trajectories
Aug 19, 2022Yaosen Min, Ye Wei, Peizhuo Wang, Nian Wu, Stefan Bauer, Shuxin Zheng, Yu Shi, Yingheng Wang, Dan Zhao, Ji Wu, Jianyang Zeng
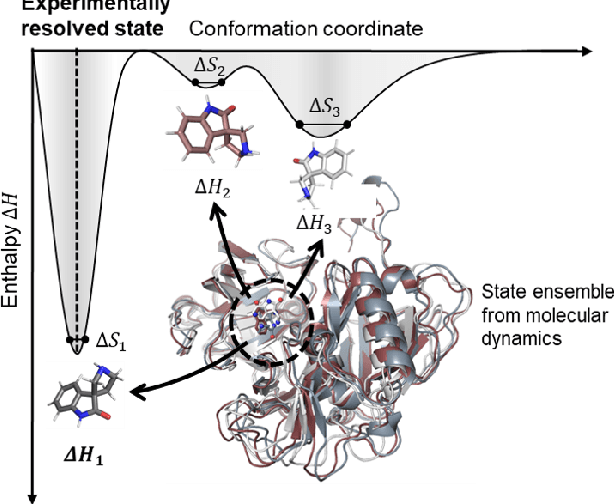
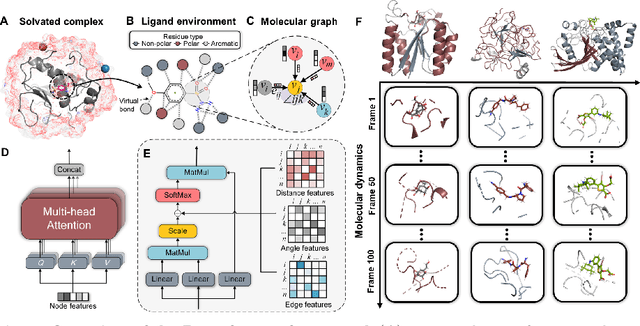
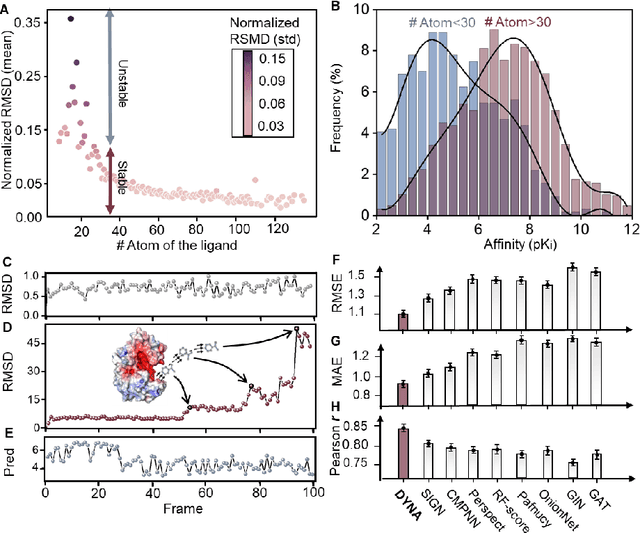

The accurate protein-ligand binding affinity prediction is essential in drug design and many other molecular recognition problems. Despite many advances in affinity prediction based on machine learning techniques, they are still limited since the protein-ligand binding is determined by the dynamics of atoms and molecules. To this end, we curated an MD dataset containing 3,218 dynamic protein-ligand complexes and further developed Dynaformer, a graph-based deep learning framework. Dynaformer can fully capture the dynamic binding rules by considering various geometric characteristics of the interaction. Our method shows superior performance over the methods hitherto reported. Moreover, we performed virtual screening on heat shock protein 90 (HSP90) by integrating our model with structure-based docking. We benchmarked our performance against other baselines, demonstrating that our method can identify the molecule with the highest experimental potency. We anticipate that large-scale MD dataset and machine learning models will form a new synergy, providing a new route towards accelerated drug discovery and optimization.
Hybrid Atlas Building with Deep Registration Priors
Dec 13, 2021Nian Wu, Jian Wang, Miaomiao Zhang, Guixu Zhang, Yaxin Peng, Chaomin Shen
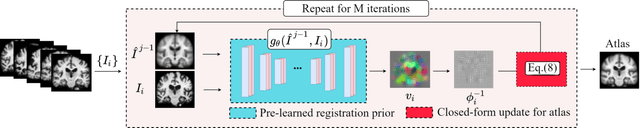
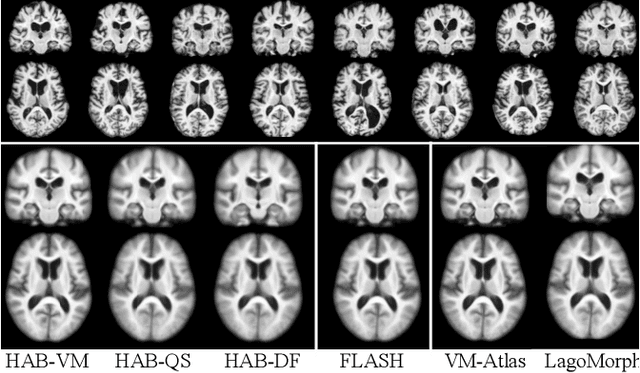
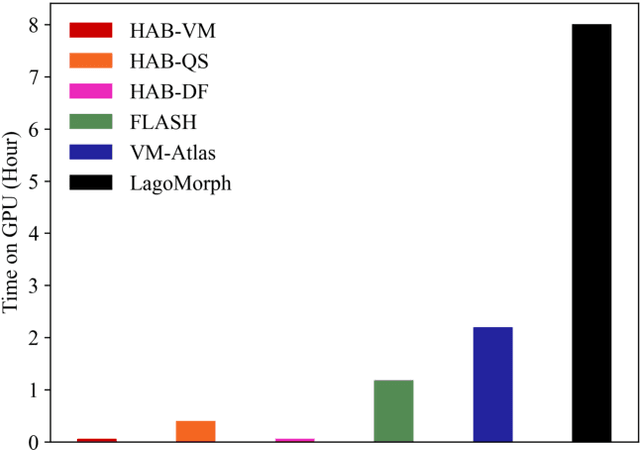
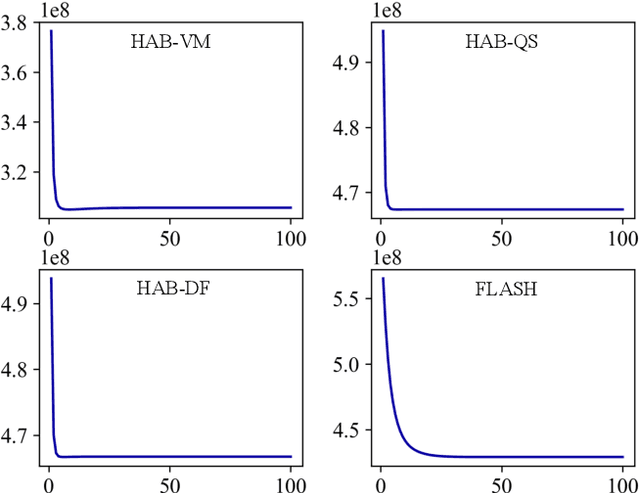
Registration-based atlas building often poses computational challenges in high-dimensional image spaces. In this paper, we introduce a novel hybrid atlas building algorithm that fast estimates atlas from large-scale image datasets with much reduced computational cost. In contrast to previous approaches that iteratively perform registration tasks between an estimated atlas and individual images, we propose to use learned priors of registration from pre-trained neural networks. This newly developed hybrid framework features several advantages of (i) providing an efficient way of atlas building without losing the quality of results, and (ii) offering flexibility in utilizing a wide variety of deep learning based registration methods. We demonstrate the effectiveness of this proposed model on 3D brain magnetic resonance imaging (MRI) scans.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge