Distributed Gesture Controlled Systems for Human-Machine Interface
Apr 12, 2023Hans Johnson, Jafar Saniie




This paper presents the design flow of an IoT human machine touchless interface. The device uses embedded computing in conjunction with the Leap Motion Controller to provide an accurate and intuitive touchless interface. Its main function is to augment current touchscreen devices in public spaces through a combination of computer vision technology, event driven programming, and machine learning. Especially following the COVID 19 pandemic, this technology is important for hygiene and sanitation purposes for public devices such as airports, food, and ATM kiosks where hundreds or even thousands of people may touch these devices in a single day. A prototype of the touchless interface was designed with a Leap Motion Controller housed on a Windows PC exchanging information with a Raspberry Pi microcontroller via internet connection.
MONAI: An open-source framework for deep learning in healthcare
Nov 04, 2022M. Jorge Cardoso, Wenqi Li, Richard Brown, Nic Ma, Eric Kerfoot, Yiheng Wang, Benjamin Murrey, Andriy Myronenko, Can Zhao, Dong Yang, Vishwesh Nath, Yufan He, Ziyue Xu, Ali Hatamizadeh, Wentao Zhu, Yun Liu, Mingxin Zheng, Yucheng Tang, Isaac Yang, Michael Zephyr, Behrooz Hashemian, Sachidanand Alle, Mohammad Zalbagi Darestani, Charlie Budd, Marc Modat, Tom Vercauteren, Guotai Wang, Yiwen Li, Yipeng Hu, Yunguan Fu, Benjamin Gorman, Hans Johnson, Brad Genereaux, Barbaros S. Erdal, Vikash Gupta, Andres Diaz-Pinto, Andre Dourson, Lena Maier-Hein, Paul F. Jaeger, Michael Baumgartner, Jayashree Kalpathy-Cramer, Mona Flores, Justin Kirby, Lee A. D. Cooper, Holger R. Roth, Daguang Xu, David Bericat, Ralf Floca, S. Kevin Zhou, Haris Shuaib, Keyvan Farahani, Klaus H. Maier-Hein, Stephen Aylward, Prerna Dogra, Sebastien Ourselin, Andrew Feng

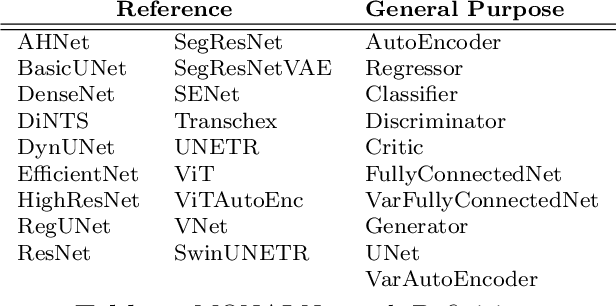
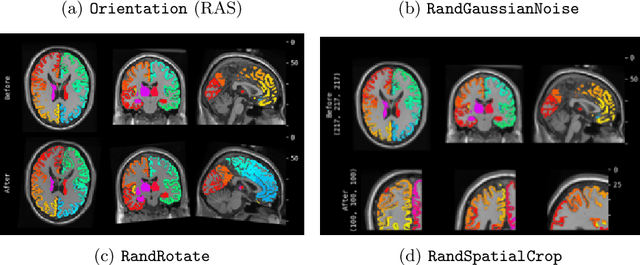
Artificial Intelligence (AI) is having a tremendous impact across most areas of science. Applications of AI in healthcare have the potential to improve our ability to detect, diagnose, prognose, and intervene on human disease. For AI models to be used clinically, they need to be made safe, reproducible and robust, and the underlying software framework must be aware of the particularities (e.g. geometry, physiology, physics) of medical data being processed. This work introduces MONAI, a freely available, community-supported, and consortium-led PyTorch-based framework for deep learning in healthcare. MONAI extends PyTorch to support medical data, with a particular focus on imaging, and provide purpose-specific AI model architectures, transformations and utilities that streamline the development and deployment of medical AI models. MONAI follows best practices for software-development, providing an easy-to-use, robust, well-documented, and well-tested software framework. MONAI preserves the simple, additive, and compositional approach of its underlying PyTorch libraries. MONAI is being used by and receiving contributions from research, clinical and industrial teams from around the world, who are pursuing applications spanning nearly every aspect of healthcare.
Tensor-Based Grading: A Novel Patch-Based Grading Approach for the Analysis of Deformation Fields in Huntington's Disease
Jan 23, 2020Kilian Hett, Hans Johnson, Pierrick Coupé, Jane Paulsen, Jeffrey Long, Ipek Oguz




The improvements in magnetic resonance imaging have led to the development of numerous techniques to better detect structural alterations caused by neurodegenerative diseases. Among these, the patch-based grading framework has been proposed to model local patterns of anatomical changes. This approach is attractive because of its low computational cost and its competitive performance. Other studies have proposed to analyze the deformations of brain structures using tensor-based morphometry, which is a highly interpretable approach. In this work, we propose to combine the advantages of these two approaches by extending the patch-based grading framework with a new tensor-based grading method that enables us to model patterns of local deformation using a log-Euclidean metric. We evaluate our new method in a study of the putamen for the classification of patients with pre-manifest Huntington's disease and healthy controls. Our experiments show a substantial increase in classification accuracy (87.5 $\pm$ 0.5 vs. 81.3 $\pm$ 0.6) compared to the existing patch-based grading methods, and a good complement to putamen volume, which is a primary imaging-based marker for the study of Huntington's disease.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge