A Robust Ensemble Algorithm for Ischemic Stroke Lesion Segmentation: Generalizability and Clinical Utility Beyond the ISLES Challenge
Apr 03, 2024Ezequiel de la Rosa, Mauricio Reyes, Sook-Lei Liew, Alexandre Hutton, Roland Wiest, Johannes Kaesmacher, Uta Hanning, Arsany Hakim, Richard Zubal, Waldo Valenzuela, David Robben, Diana M. Sima, Vincenzo Anania, Arne Brys, James A. Meakin, Anne Mickan, Gabriel Broocks, Christian Heitkamp, Shengbo Gao, Kongming Liang, Ziji Zhang, Md Mahfuzur Rahman Siddiquee, Andriy Myronenko, Pooya Ashtari, Sabine Van Huffel, Hyun-su Jeong, Chi-ho Yoon, Chulhong Kim, Jiayu Huo, Sebastien Ourselin, Rachel Sparks, Albert Clèrigues, Arnau Oliver, Xavier Lladó, Liam Chalcroft, Ioannis Pappas, Jeroen Bertels, Ewout Heylen, Juliette Moreau, Nima Hatami, Carole Frindel, Abdul Qayyum, Moona Mazher, Domenec Puig, Shao-Chieh Lin, Chun-Jung Juan, Tianxi Hu, Lyndon Boone, Maged Goubran, Yi-Jui Liu, Susanne Wegener, Florian Kofler, Ivan Ezhov, Suprosanna Shit, Moritz R. Hernandez Petzsche, Bjoern Menze, Jan S. Kirschke, Benedikt Wiestler
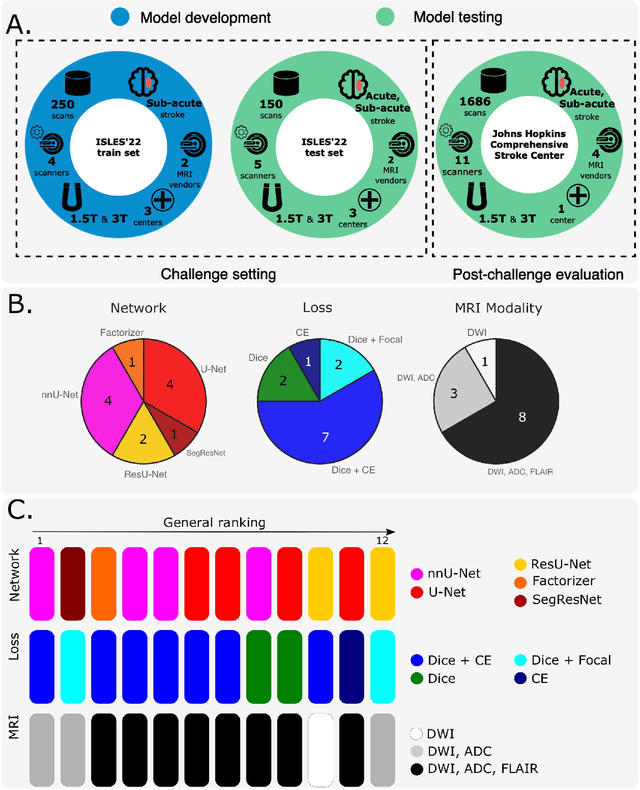
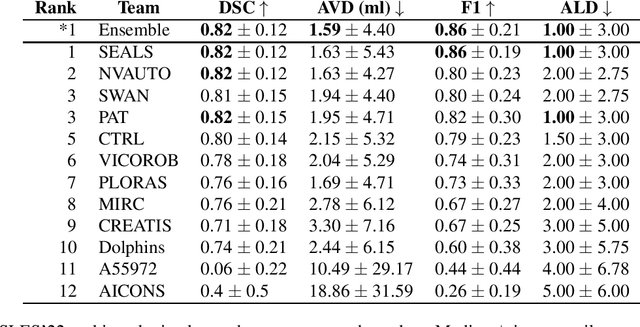
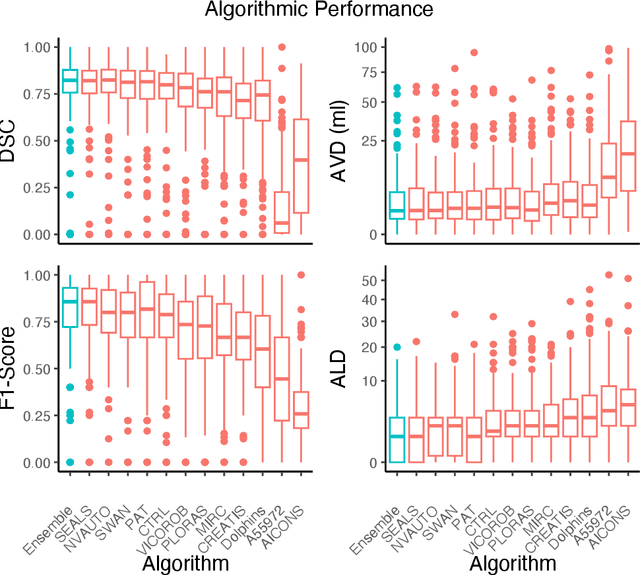
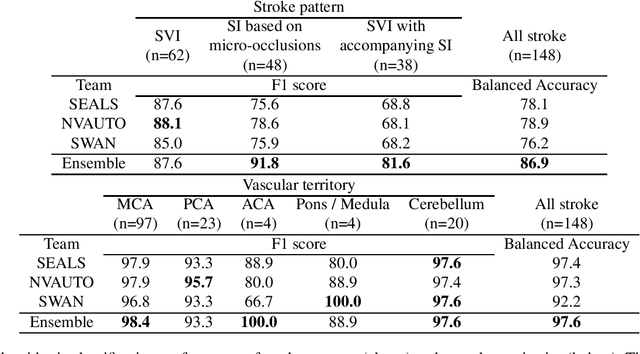
Diffusion-weighted MRI (DWI) is essential for stroke diagnosis, treatment decisions, and prognosis. However, image and disease variability hinder the development of generalizable AI algorithms with clinical value. We address this gap by presenting a novel ensemble algorithm derived from the 2022 Ischemic Stroke Lesion Segmentation (ISLES) challenge. ISLES'22 provided 400 patient scans with ischemic stroke from various medical centers, facilitating the development of a wide range of cutting-edge segmentation algorithms by the research community. Through collaboration with leading teams, we combined top-performing algorithms into an ensemble model that overcomes the limitations of individual solutions. Our ensemble model achieved superior ischemic lesion detection and segmentation accuracy on our internal test set compared to individual algorithms. This accuracy generalized well across diverse image and disease variables. Furthermore, the model excelled in extracting clinical biomarkers. Notably, in a Turing-like test, neuroradiologists consistently preferred the algorithm's segmentations over manual expert efforts, highlighting increased comprehensiveness and precision. Validation using a real-world external dataset (N=1686) confirmed the model's generalizability. The algorithm's outputs also demonstrated strong correlations with clinical scores (admission NIHSS and 90-day mRS) on par with or exceeding expert-derived results, underlining its clinical relevance. This study offers two key findings. First, we present an ensemble algorithm (https://github.com/Tabrisrei/ISLES22_Ensemble) that detects and segments ischemic stroke lesions on DWI across diverse scenarios on par with expert (neuro)radiologists. Second, we show the potential for biomedical challenge outputs to extend beyond the challenge's initial objectives, demonstrating their real-world clinical applicability.
Multi-Center Fetal Brain Tissue Annotation (FeTA) Challenge 2022 Results
Feb 08, 2024Kelly Payette, Céline Steger, Roxane Licandro, Priscille de Dumast, Hongwei Bran Li, Matthew Barkovich, Liu Li, Maik Dannecker, Chen Chen, Cheng Ouyang, Niccolò McConnell, Alina Miron, Yongmin Li, Alena Uus, Irina Grigorescu, Paula Ramirez Gilliland, Md Mahfuzur Rahman Siddiquee, Daguang Xu, Andriy Myronenko, Haoyu Wang, Ziyan Huang, Jin Ye, Mireia Alenyà, Valentin Comte, Oscar Camara, Jean-Baptiste Masson, Astrid Nilsson, Charlotte Godard, Moona Mazher, Abdul Qayyum, Yibo Gao, Hangqi Zhou, Shangqi Gao, Jia Fu, Guiming Dong, Guotai Wang, ZunHyan Rieu, HyeonSik Yang, Minwoo Lee, Szymon Płotka, Michal K. Grzeszczyk, Arkadiusz Sitek, Luisa Vargas Daza, Santiago Usma, Pablo Arbelaez, Wenying Lu, Wenhao Zhang, Jing Liang, Romain Valabregue, Anand A. Joshi, Krishna N. Nayak, Richard M. Leahy, Luca Wilhelmi, Aline Dändliker, Hui Ji, Antonio G. Gennari, Anton Jakovčić, Melita Klaić, Ana Adžić, Pavel Marković, Gracia Grabarić, Gregor Kasprian, Gregor Dovjak, Milan Rados, Lana Vasung, Meritxell Bach Cuadra, Andras Jakab
Segmentation is a critical step in analyzing the developing human fetal brain. There have been vast improvements in automatic segmentation methods in the past several years, and the Fetal Brain Tissue Annotation (FeTA) Challenge 2021 helped to establish an excellent standard of fetal brain segmentation. However, FeTA 2021 was a single center study, and the generalizability of algorithms across different imaging centers remains unsolved, limiting real-world clinical applicability. The multi-center FeTA Challenge 2022 focuses on advancing the generalizability of fetal brain segmentation algorithms for magnetic resonance imaging (MRI). In FeTA 2022, the training dataset contained images and corresponding manually annotated multi-class labels from two imaging centers, and the testing data contained images from these two imaging centers as well as two additional unseen centers. The data from different centers varied in many aspects, including scanners used, imaging parameters, and fetal brain super-resolution algorithms applied. 16 teams participated in the challenge, and 17 algorithms were evaluated. Here, a detailed overview and analysis of the challenge results are provided, focusing on the generalizability of the submissions. Both in- and out of domain, the white matter and ventricles were segmented with the highest accuracy, while the most challenging structure remains the cerebral cortex due to anatomical complexity. The FeTA Challenge 2022 was able to successfully evaluate and advance generalizability of multi-class fetal brain tissue segmentation algorithms for MRI and it continues to benchmark new algorithms. The resulting new methods contribute to improving the analysis of brain development in utero.
Aorta Segmentation from 3D CT in MICCAI SEG.A. 2023 Challenge
Oct 06, 2023Andriy Myronenko, Dong Yang, Yufan He, Daguang Xu
Aorta provides the main blood supply of the body. Screening of aorta with imaging helps for early aortic disease detection and monitoring. In this work, we describe our solution to the Segmentation of the Aorta (SEG.A.231) from 3D CT challenge. We use automated segmentation method Auto3DSeg available in MONAI. Our solution achieves an average Dice score of 0.920 and 95th percentile of the Hausdorff Distance (HD95) of 6.013, which ranks first and wins the SEG.A. 2023 challenge.
Automated 3D Segmentation of Kidneys and Tumors in MICCAI KiTS 2023 Challenge
Oct 06, 2023Andriy Myronenko, Dong Yang, Yufan He, Daguang Xu
Kidney and Kidney Tumor Segmentation Challenge (KiTS) 2023 offers a platform for researchers to compare their solutions to segmentation from 3D CT. In this work, we describe our submission to the challenge using automated segmentation of Auto3DSeg available in MONAI. Our solution achieves the average dice of 0.835 and surface dice of 0.723, which ranks first and wins the KiTS 2023 challenge.
The state-of-the-art 3D anisotropic intracranial hemorrhage segmentation on non-contrast head CT: The INSTANCE challenge
Jan 12, 2023Xiangyu Li, Gongning Luo, Kuanquan Wang, Hongyu Wang, Jun Liu, Xinjie Liang, Jie Jiang, Zhenghao Song, Chunyue Zheng, Haokai Chi, Mingwang Xu, Yingte He, Xinghua Ma, Jingwen Guo, Yifan Liu, Chuanpu Li, Zeli Chen, Md Mahfuzur Rahman Siddiquee, Andriy Myronenko, Antoine P. Sanner, Anirban Mukhopadhyay, Ahmed E. Othman, Xingyu Zhao, Weiping Liu, Jinhuang Zhang, Xiangyuan Ma, Qinghui Liu, Bradley J. MacIntosh, Wei Liang, Moona Mazher, Abdul Qayyum, Valeriia Abramova, Xavier Lladó, Shuo Li
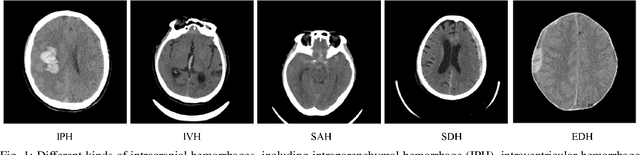

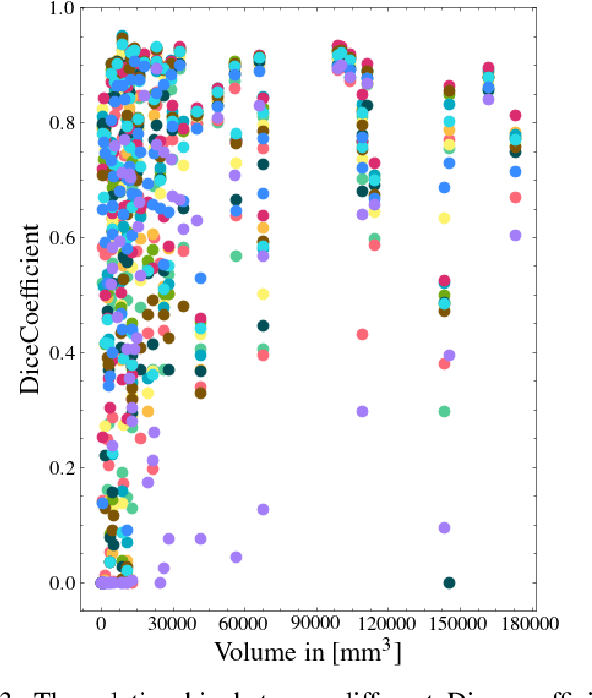
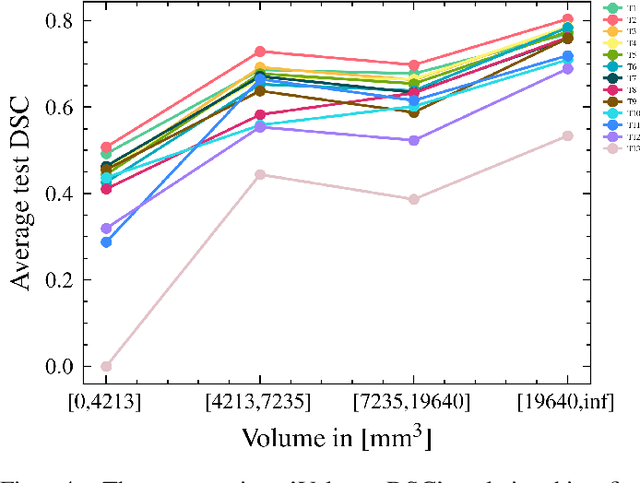
Automatic intracranial hemorrhage segmentation in 3D non-contrast head CT (NCCT) scans is significant in clinical practice. Existing hemorrhage segmentation methods usually ignores the anisotropic nature of the NCCT, and are evaluated on different in-house datasets with distinct metrics, making it highly challenging to improve segmentation performance and perform objective comparisons among different methods. The INSTANCE 2022 was a grand challenge held in conjunction with the 2022 International Conference on Medical Image Computing and Computer Assisted Intervention (MICCAI). It is intended to resolve the above-mentioned problems and promote the development of both intracranial hemorrhage segmentation and anisotropic data processing. The INSTANCE released a training set of 100 cases with ground-truth and a validation set with 30 cases without ground-truth labels that were available to the participants. A held-out testing set with 70 cases is utilized for the final evaluation and ranking. The methods from different participants are ranked based on four metrics, including Dice Similarity Coefficient (DSC), Hausdorff Distance (HD), Relative Volume Difference (RVD) and Normalized Surface Dice (NSD). A total of 13 teams submitted distinct solutions to resolve the challenges, making several baseline models, pre-processing strategies and anisotropic data processing techniques available to future researchers. The winner method achieved an average DSC of 0.6925, demonstrating a significant growth over our proposed baseline method. To the best of our knowledge, the proposed INSTANCE challenge releases the first intracranial hemorrhage segmentation benchmark, and is also the first challenge that intended to resolve the anisotropic problem in 3D medical image segmentation, which provides new alternatives in these research fields.
MONAI: An open-source framework for deep learning in healthcare
Nov 04, 2022M. Jorge Cardoso, Wenqi Li, Richard Brown, Nic Ma, Eric Kerfoot, Yiheng Wang, Benjamin Murrey, Andriy Myronenko, Can Zhao, Dong Yang, Vishwesh Nath, Yufan He, Ziyue Xu, Ali Hatamizadeh, Wentao Zhu, Yun Liu, Mingxin Zheng, Yucheng Tang, Isaac Yang, Michael Zephyr, Behrooz Hashemian, Sachidanand Alle, Mohammad Zalbagi Darestani, Charlie Budd, Marc Modat, Tom Vercauteren, Guotai Wang, Yiwen Li, Yipeng Hu, Yunguan Fu, Benjamin Gorman, Hans Johnson, Brad Genereaux, Barbaros S. Erdal, Vikash Gupta, Andres Diaz-Pinto, Andre Dourson, Lena Maier-Hein, Paul F. Jaeger, Michael Baumgartner, Jayashree Kalpathy-Cramer, Mona Flores, Justin Kirby, Lee A. D. Cooper, Holger R. Roth, Daguang Xu, David Bericat, Ralf Floca, S. Kevin Zhou, Haris Shuaib, Keyvan Farahani, Klaus H. Maier-Hein, Stephen Aylward, Prerna Dogra, Sebastien Ourselin, Andrew Feng

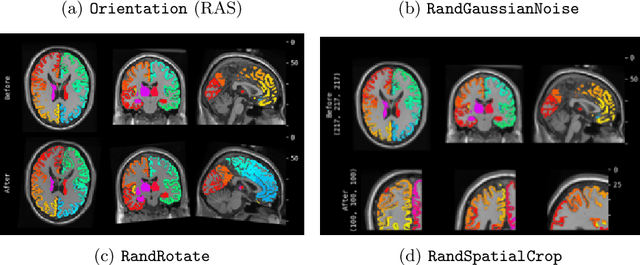
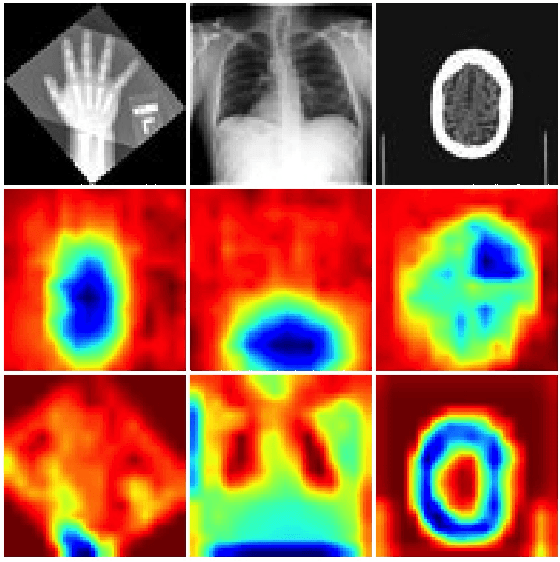
Artificial Intelligence (AI) is having a tremendous impact across most areas of science. Applications of AI in healthcare have the potential to improve our ability to detect, diagnose, prognose, and intervene on human disease. For AI models to be used clinically, they need to be made safe, reproducible and robust, and the underlying software framework must be aware of the particularities (e.g. geometry, physiology, physics) of medical data being processed. This work introduces MONAI, a freely available, community-supported, and consortium-led PyTorch-based framework for deep learning in healthcare. MONAI extends PyTorch to support medical data, with a particular focus on imaging, and provide purpose-specific AI model architectures, transformations and utilities that streamline the development and deployment of medical AI models. MONAI follows best practices for software-development, providing an easy-to-use, robust, well-documented, and well-tested software framework. MONAI preserves the simple, additive, and compositional approach of its underlying PyTorch libraries. MONAI is being used by and receiving contributions from research, clinical and industrial teams from around the world, who are pursuing applications spanning nearly every aspect of healthcare.
NVIDIA FLARE: Federated Learning from Simulation to Real-World
Oct 24, 2022Holger R. Roth, Yan Cheng, Yuhong Wen, Isaac Yang, Ziyue Xu, Yuan-Ting Hsieh, Kristopher Kersten, Ahmed Harouni, Can Zhao, Kevin Lu, Zhihong Zhang, Wenqi Li, Andriy Myronenko, Dong Yang, Sean Yang, Nicola Rieke, Abood Quraini, Chester Chen, Daguang Xu, Nic Ma, Prerna Dogra, Mona Flores, Andrew Feng
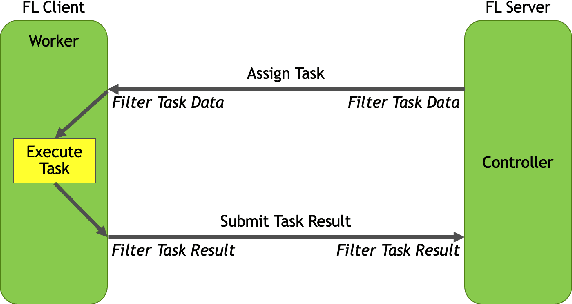
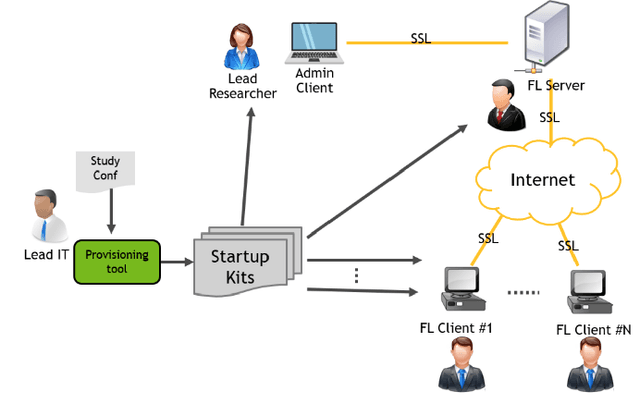
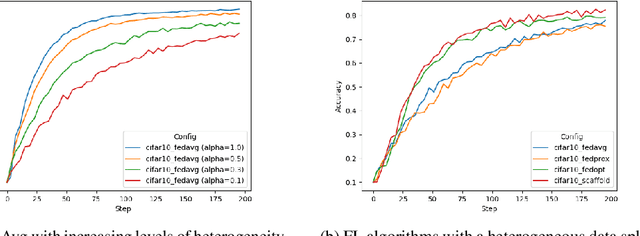

Federated learning (FL) enables building robust and generalizable AI models by leveraging diverse datasets from multiple collaborators without centralizing the data. We created NVIDIA FLARE as an open-source software development kit (SDK) to make it easier for data scientists to use FL in their research and real-world applications. The SDK includes solutions for state-of-the-art FL algorithms and federated machine learning approaches, which facilitate building workflows for distributed learning across enterprises and enable platform developers to create a secure, privacy-preserving offering for multiparty collaboration utilizing homomorphic encryption or differential privacy. The SDK is a lightweight, flexible, and scalable Python package, and allows researchers to bring their data science workflows implemented in any training libraries (PyTorch, TensorFlow, XGBoost, or even NumPy) and apply them in real-world FL settings. This paper introduces the key design principles of FLARE and illustrates some use cases (e.g., COVID analysis) with customizable FL workflows that implement different privacy-preserving algorithms. Code is available at https://github.com/NVIDIA/NVFlare.
Automated head and neck tumor segmentation from 3D PET/CT
Sep 22, 2022Andriy Myronenko, Md Mahfuzur Rahman Siddiquee, Dong Yang, Yufan He, Daguang Xu
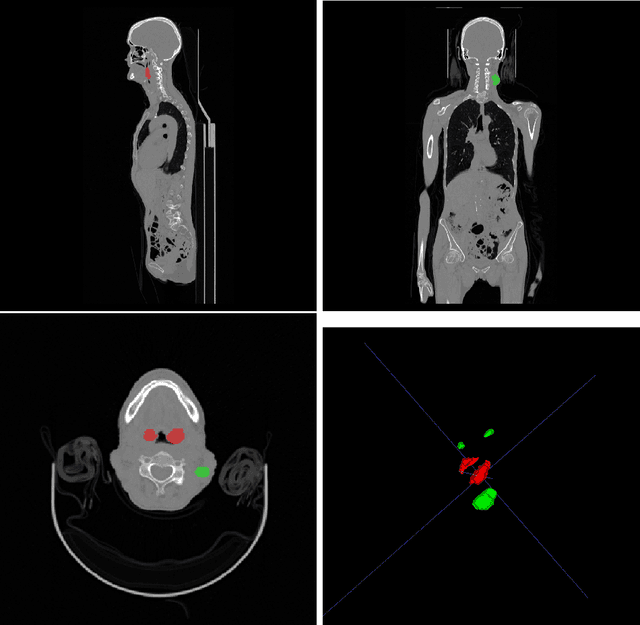
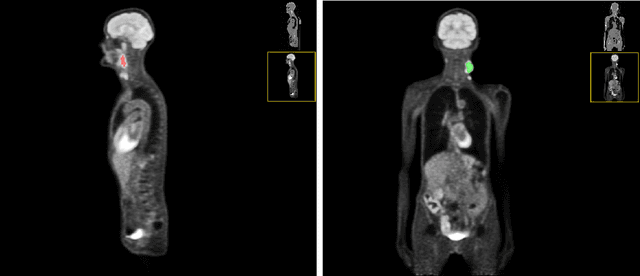
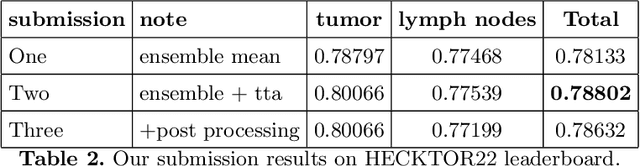
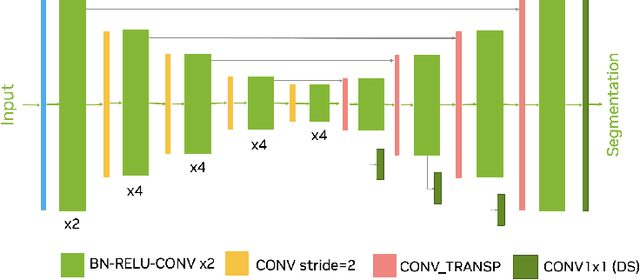
Head and neck tumor segmentation challenge (HECKTOR) 2022 offers a platform for researchers to compare their solutions to segmentation of tumors and lymph nodes from 3D CT and PET images. In this work, we describe our solution to HECKTOR 2022 segmentation task. We re-sample all images to a common resolution, crop around head and neck region, and train SegResNet semantic segmentation network from MONAI. We use 5-fold cross validation to select best model checkpoints. The final submission is an ensemble of 15 models from 3 runs. Our solution (team name NVAUTO) achieves the 1st place on the HECKTOR22 challenge leaderboard with an aggregated dice score of 0.78802.
Automated segmentation of intracranial hemorrhages from 3D CT
Sep 21, 2022Md Mahfuzur Rahman Siddiquee, Dong Yang, Yufan He, Daguang Xu, Andriy Myronenko
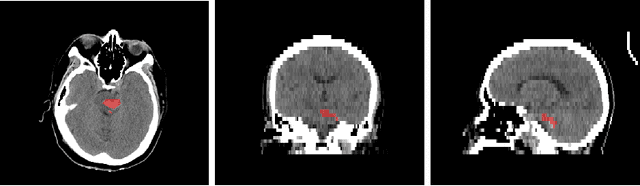
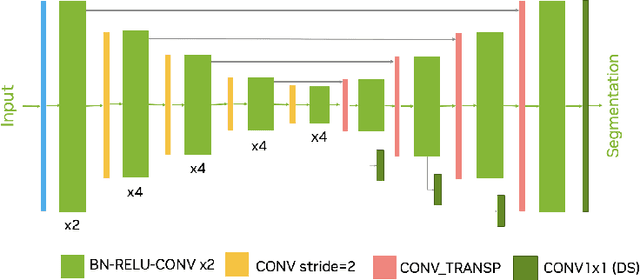
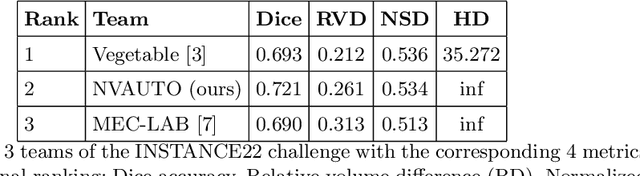
Intracranial hemorrhage segmentation challenge (INSTANCE 2022) offers a platform for researchers to compare their solutions to segmentation of hemorrhage stroke regions from 3D CTs. In this work, we describe our solution to INSTANCE 2022. We use a 2D segmentation network, SegResNet from MONAI, operating slice-wise without resampling. The final submission is an ensemble of 18 models. Our solution (team name NVAUTO) achieves the top place in terms of Dice metric (0.721), and overall rank 2. It is implemented with Auto3DSeg.
Automated ischemic stroke lesion segmentation from 3D MRI
Sep 21, 2022Md Mahfuzur Rahman Siddique, Dong Yang, Yufan He, Daguang Xu, Andriy Myronenko
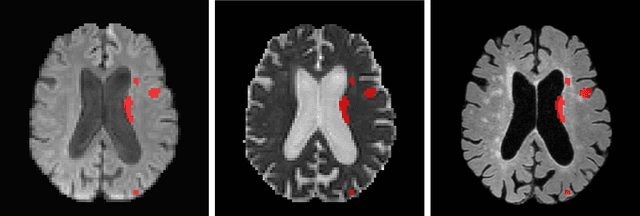
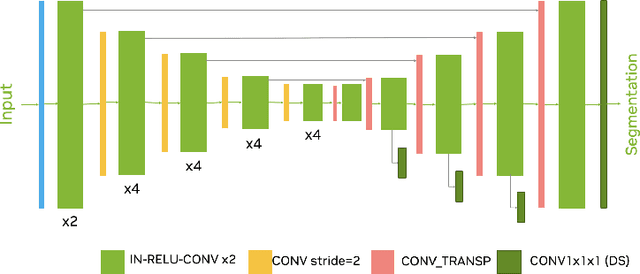
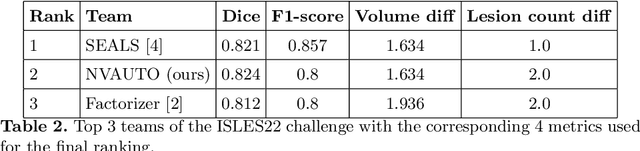
Ischemic Stroke Lesion Segmentation challenge (ISLES 2022) offers a platform for researchers to compare their solutions to 3D segmentation of ischemic stroke regions from 3D MRIs. In this work, we describe our solution to ISLES 2022 segmentation task. We re-sample all images to a common resolution, use two input MRI modalities (DWI and ADC) and train SegResNet semantic segmentation network from MONAI. The final submission is an ensemble of 15 models (from 3 runs of 5-fold cross validation). Our solution (team name NVAUTO) achieves the top place in terms of Dice metric (0.824), and overall rank 2 (based on the combined metric ranking).
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge