RESTORE: Towards Feature Shift for Vision-Language Prompt Learning
Mar 10, 2024Yuncheng Yang, Chuyan Zhang, Zuopeng Yang, Yuting Gao, Yulei Qin, Ke Li, Xing Sun, Jie Yang, Yun Gu
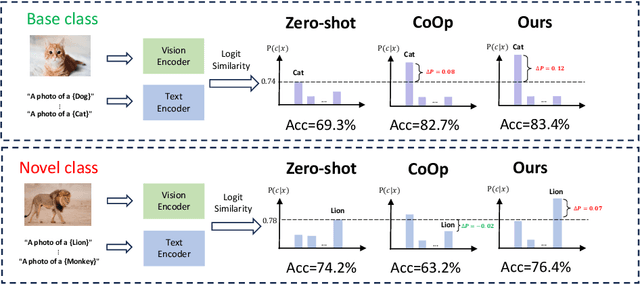
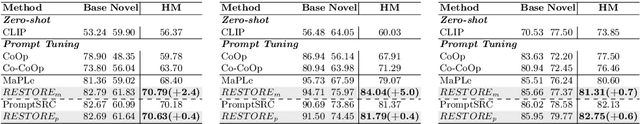
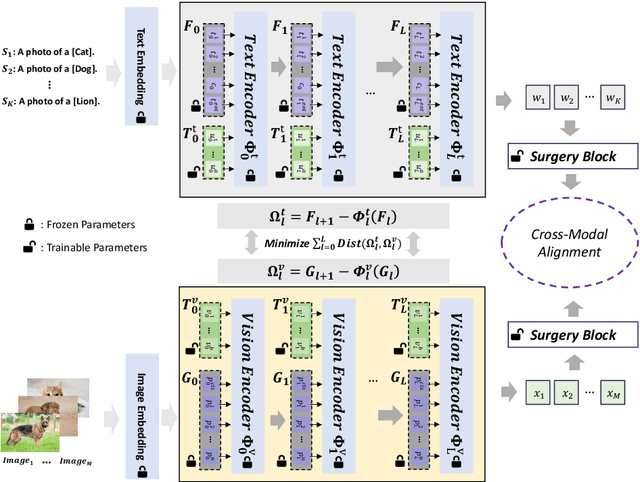
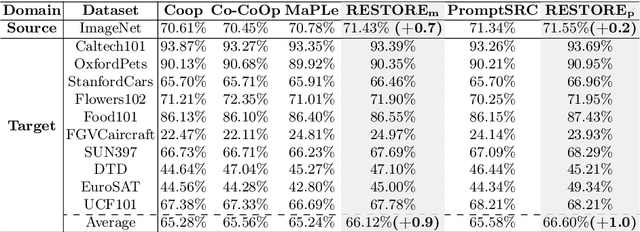
Prompt learning is effective for fine-tuning foundation models to improve their generalization across a variety of downstream tasks. However, the prompts that are independently optimized along a single modality path, may sacrifice the vision-language alignment of pre-trained models in return for improved performance on specific tasks and classes, leading to poorer generalization. In this paper, we first demonstrate that prompt tuning along only one single branch of CLIP (e.g., language or vision) is the reason why the misalignment occurs. Without proper regularization across the learnable parameters in different modalities, prompt learning violates the original pre-training constraints inherent in the two-tower architecture. To address such misalignment, we first propose feature shift, which is defined as the variation of embeddings after introducing the learned prompts, to serve as an explanatory tool. We dive into its relation with generalizability and thereafter propose RESTORE, a multi-modal prompt learning method that exerts explicit constraints on cross-modal consistency. To be more specific, to prevent feature misalignment, a feature shift consistency is introduced to synchronize inter-modal feature shifts by measuring and regularizing the magnitude of discrepancy during prompt tuning. In addition, we propose a "surgery" block to avoid short-cut hacking, where cross-modal misalignment can still be severe if the feature shift of each modality varies drastically at the same rate. It is implemented as feed-forward adapters upon both modalities to alleviate the misalignment problem. Extensive experiments on 15 datasets demonstrate that our method outperforms the state-of-the-art prompt tuning methods without compromising feature alignment.
AC-Norm: Effective Tuning for Medical Image Analysis via Affine Collaborative Normalization
Jul 28, 2023Chuyan Zhang, Yuncheng Yang, Hao Zheng, Yun Gu
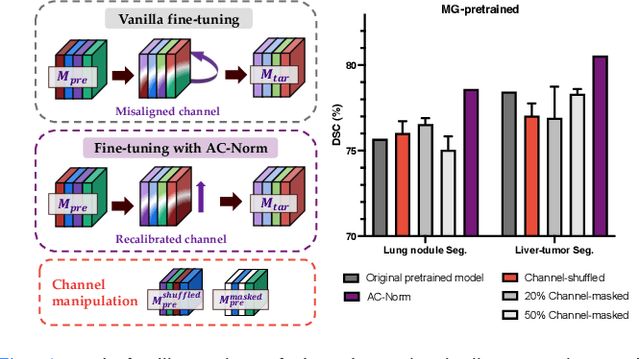
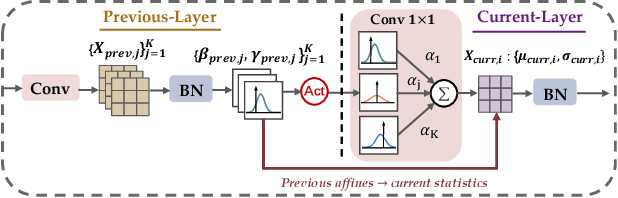
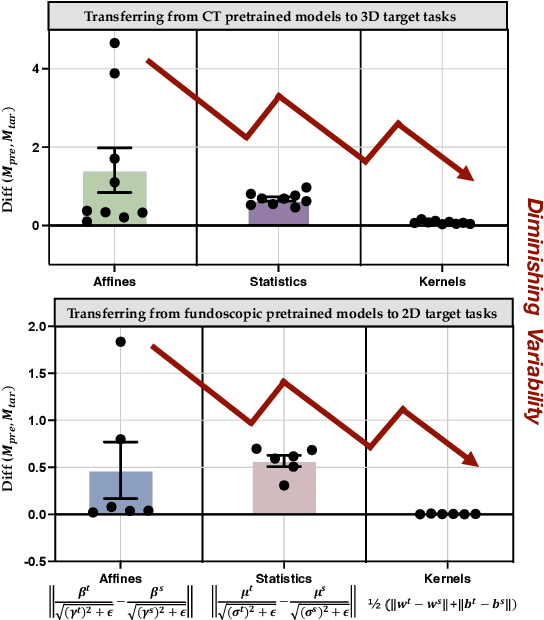
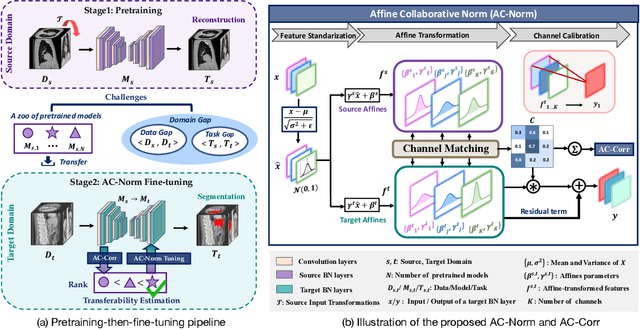
Driven by the latest trend towards self-supervised learning (SSL), the paradigm of "pretraining-then-finetuning" has been extensively explored to enhance the performance of clinical applications with limited annotations. Previous literature on model finetuning has mainly focused on regularization terms and specific policy models, while the misalignment of channels between source and target models has not received sufficient attention. In this work, we revisited the dynamics of batch normalization (BN) layers and observed that the trainable affine parameters of BN serve as sensitive indicators of domain information. Therefore, Affine Collaborative Normalization (AC-Norm) is proposed for finetuning, which dynamically recalibrates the channels in the target model according to the cross-domain channel-wise correlations without adding extra parameters. Based on a single-step backpropagation, AC-Norm can also be utilized to measure the transferability of pretrained models. We evaluated AC-Norm against the vanilla finetuning and state-of-the-art fine-tuning methods on transferring diverse pretrained models to the diabetic retinopathy grade classification, retinal vessel segmentation, CT lung nodule segmentation/classification, CT liver-tumor segmentation and MRI cardiac segmentation tasks. Extensive experiments demonstrate that AC-Norm unanimously outperforms the vanilla finetuning by up to 4% improvement, even under significant domain shifts where the state-of-the-art methods bring no gains. We also prove the capability of AC-Norm in fast transferability estimation. Our code is available at https://github.com/EndoluminalSurgicalVision-IMR/ACNorm.
Dive into Self-Supervised Learning for Medical Image Analysis: Data, Models and Tasks
Sep 25, 2022Chuyan Zhang, Yun Gu
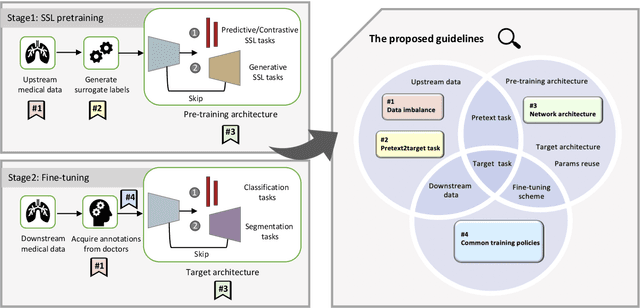
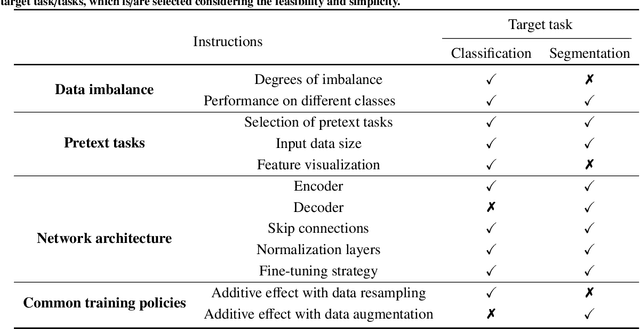
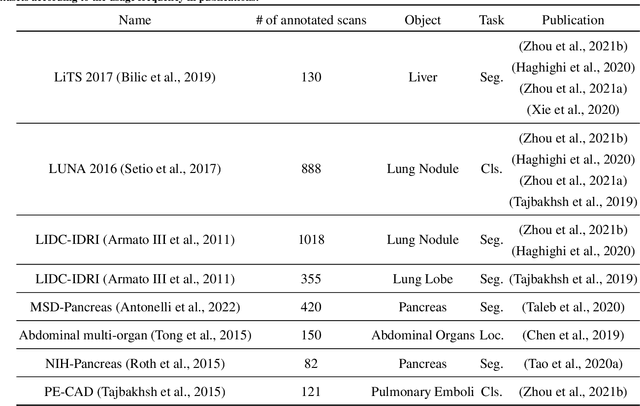
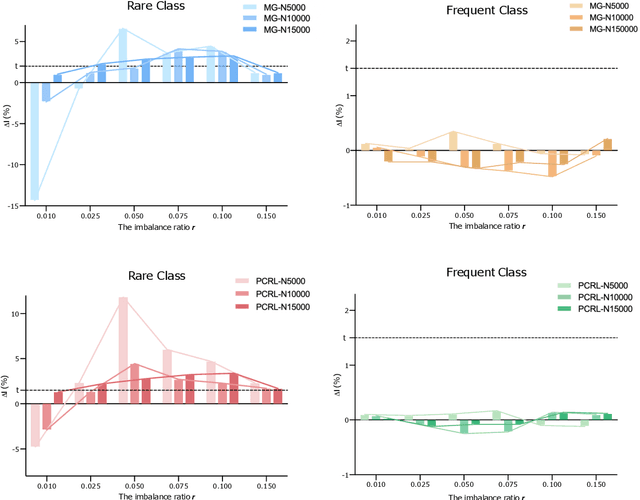
Self-supervised learning (SSL) has achieved remarkable performance on various medical imaging tasks by dint of priors from massive unlabeled data. However, for a specific downstream task, there is still a lack of an instruction book on how to select suitable pretext tasks and implementation details. In this work, we first review the latest applications of self-supervised methods in the field of medical imaging analysis. Then, we conduct extensive experiments to explore four significant issues in SSL for medical imaging, including (1) the effect of self-supervised pretraining on imbalanced datasets, (2) network architectures, (3) the applicability of upstream tasks to downstream tasks and (4) the stacking effect of SSL and commonly used policies for deep learning, including data resampling and augmentation. Based on the experimental results, potential guidelines are presented for self-supervised pretraining in medical imaging. Finally, we discuss future research directions and raise issues to be aware of when designing new SSL methods and paradigms.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge