Hunting imaging biomarkers in pulmonary fibrosis: Benchmarks of the AIIB23 challenge
Dec 21, 2023Yang Nan, Xiaodan Xing, Shiyi Wang, Zeyu Tang, Federico N Felder, Sheng Zhang, Roberta Eufrasia Ledda, Xiaoliu Ding, Ruiqi Yu, Weiping Liu, Feng Shi, Tianyang Sun, Zehong Cao, Minghui Zhang, Yun Gu, Hanxiao Zhang, Jian Gao, Wen Tang, Pengxin Yu, Han Kang, Junqiang Chen, Xing Lu, Boyu Zhang, Michail Mamalakis, Francesco Prinzi, Gianluca Carlini, Lisa Cuneo, Abhirup Banerjee, Zhaohu Xing, Lei Zhu, Zacharia Mesbah, Dhruv Jain, Tsiry Mayet, Hongyu Yuan, Qing Lyu, Athol Wells, Simon LF Walsh, Guang Yang
Airway-related quantitative imaging biomarkers are crucial for examination, diagnosis, and prognosis in pulmonary diseases. However, the manual delineation of airway trees remains prohibitively time-consuming. While significant efforts have been made towards enhancing airway modelling, current public-available datasets concentrate on lung diseases with moderate morphological variations. The intricate honeycombing patterns present in the lung tissues of fibrotic lung disease patients exacerbate the challenges, often leading to various prediction errors. To address this issue, the 'Airway-Informed Quantitative CT Imaging Biomarker for Fibrotic Lung Disease 2023' (AIIB23) competition was organized in conjunction with the official 2023 International Conference on Medical Image Computing and Computer Assisted Intervention (MICCAI). The airway structures were meticulously annotated by three experienced radiologists. Competitors were encouraged to develop automatic airway segmentation models with high robustness and generalization abilities, followed by exploring the most correlated QIB of mortality prediction. A training set of 120 high-resolution computerised tomography (HRCT) scans were publicly released with expert annotations and mortality status. The online validation set incorporated 52 HRCT scans from patients with fibrotic lung disease and the offline test set included 140 cases from fibrosis and COVID-19 patients. The results have shown that the capacity of extracting airway trees from patients with fibrotic lung disease could be enhanced by introducing voxel-wise weighted general union loss and continuity loss. In addition to the competitive image biomarkers for prognosis, a strong airway-derived biomarker (Hazard ratio>1.5, p<0.0001) was revealed for survival prognostication compared with existing clinical measurements, clinician assessment and AI-based biomarkers.
A novel framework employing deep multi-attention channels network for the autonomous detection of metastasizing cells through fluorescence microscopy
Sep 02, 2023Michail Mamalakis, Sarah C. Macfarlane, Scott V. Notley, Annica K. B Gad, George Panoutsos

We developed a transparent computational large-scale imaging-based framework that can distinguish between normal and metastasizing human cells. The method relies on fluorescence microscopy images showing the spatial organization of actin and vimentin filaments in normal and metastasizing single cells, using a combination of multi-attention channels network and global explainable techniques. We test a classification between normal cells (Bj primary fibroblast), and their isogenically matched, transformed and invasive counterpart (BjTertSV40TRasV12). Manual annotation is not trivial to automate due to the intricacy of the biologically relevant features. In this research, we utilized established deep learning networks and our new multi-attention channel architecture. To increase the interpretability of the network - crucial for this application area - we developed an interpretable global explainable approach correlating the weighted geometric mean of the total cell images and their local GradCam scores. The significant results from our analysis unprecedently allowed a more detailed, and biologically relevant understanding of the cytoskeletal changes that accompany oncogenic transformation of normal to invasive and metastasizing cells. We also paved the way for a possible spatial micrometre-level biomarker for future development of diagnostic tools against metastasis (spatial distribution of vimentin).
A 3D explainability framework to uncover learning patterns and crucial sub-regions in variable sulci recognition
Sep 02, 2023Michail Mamalakis, Heloise de Vareilles, Atheer AI-Manea, Samantha C. Mitchell, Ingrid Arartz, Lynn Egeland Morch-Johnsen, Jane Garrison, Jon Simons, Pietro Lio, John Suckling, Graham Murray
Precisely identifying sulcal features in brain MRI is made challenging by the variability of brain folding. This research introduces an innovative 3D explainability frame-work that validates outputs from deep learning networks in their ability to detect the paracingulate sulcus, an anatomical feature that may or may not be present on the frontal medial surface of the human brain. This study trained and tested two networks, amalgamating local explainability techniques GradCam and SHAP with a dimensionality reduction method. The explainability framework provided both localized and global explanations, along with accuracy of classification results, revealing pertinent sub-regions contributing to the decision process through a post-fusion transformation of explanatory and statistical features. Leveraging the TOP-OSLO dataset of MRI acquired from patients with schizophrenia, greater accuracies of paracingulate sulcus detection (presence or absence) were found in the left compared to right hemispheres with distinct, but extensive sub-regions contributing to each classification outcome. The study also inadvertently highlighted the critical role of an unbiased annotation protocol in maintaining network performance fairness. Our proposed method not only offers automated, impartial annotations of a variable sulcus but also provides insights into the broader anatomical variations associated with its presence throughout the brain. The adoption of this methodology holds promise for instigating further explorations and inquiries in the field of neuroscience.
DenResCov-19: A deep transfer learning network for robust automatic classification of COVID-19, pneumonia, and tuberculosis from X-rays
Apr 08, 2021Michail Mamalakis, Andrew J. Swift, Bart Vorselaars, Surajit Ray, Simonne Weeks, Weiping Ding, Richard H. Clayton, Louise S. Mackenzie, Abhirup Banerjee
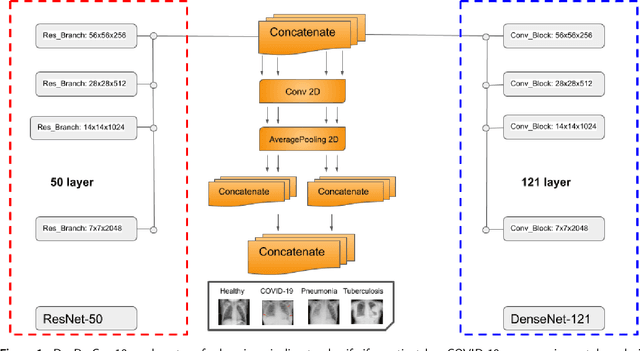



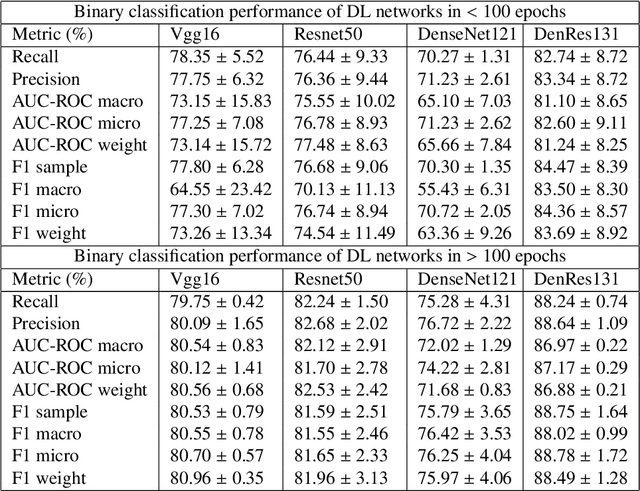
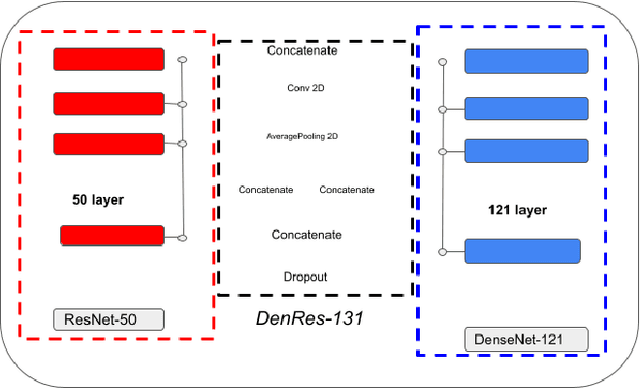
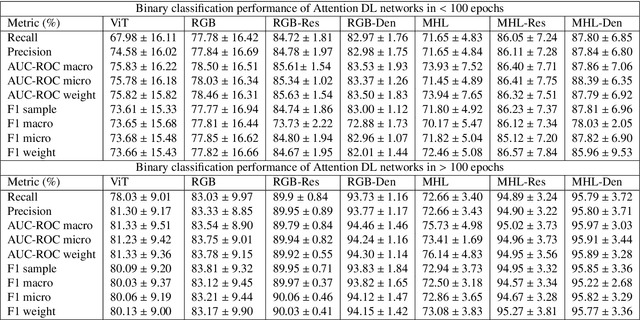
The global pandemic of COVID-19 is continuing to have a significant effect on the well-being of global population, increasing the demand for rapid testing, diagnosis, and treatment. Along with COVID-19, other etiologies of pneumonia and tuberculosis constitute additional challenges to the medical system. In this regard, the objective of this work is to develop a new deep transfer learning pipeline to diagnose patients with COVID-19, pneumonia, and tuberculosis, based on chest x-ray images. We observed in some instances DenseNet and Resnet have orthogonal performances. In our proposed model, we have created an extra layer with convolutional neural network blocks to combine these two models to establish superior performance over either model. The same strategy can be useful in other applications where two competing networks with complementary performance are observed. We have tested the performance of our proposed network on two-class (pneumonia vs healthy), three-class (including COVID-19), and four-class (including tuberculosis) classification problems. The proposed network has been able to successfully classify these lung diseases in all four datasets and has provided significant improvement over the benchmark networks of DenseNet, ResNet, and Inception-V3. These novel findings can deliver a state-of-the-art pre-screening fast-track decision network to detect COVID-19 and other lung pathologies.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge