Asymmetric and trial-dependent modeling: the contribution of LIA to SdSV Challenge Task 2
Mar 28, 2024Pierre-Michel Bousquet, Mickael Rouvier

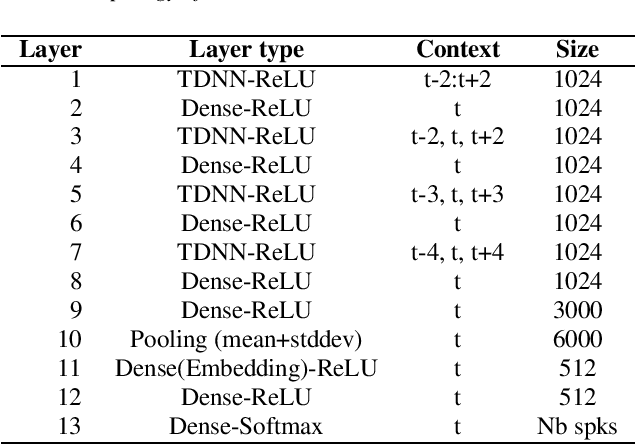


The SdSv challenge Task 2 provided an opportunity to assess efficiency and robustness of modern text-independent speaker verification systems. But it also made it possible to test new approaches, capable of taking into account the main issues of this challenge (duration, language, ...). This paper describes the contributions of our laboratory to the speaker recognition field. These contributions highlight two other challenges in addition to short-duration and language: the mismatch between enrollment and test data and the one between subsets of the evaluation trial dataset. The proposed approaches experimentally show their relevance and efficiency on the SdSv evaluation, and could be of interest in many real-life applications.
Probing the Information Encoded in Neural-based Acoustic Models of Automatic Speech Recognition Systems
Feb 29, 2024Quentin Raymondaud, Mickael Rouvier, Richard Dufour
Deep learning architectures have made significant progress in terms of performance in many research areas. The automatic speech recognition (ASR) field has thus benefited from these scientific and technological advances, particularly for acoustic modeling, now integrating deep neural network architectures. However, these performance gains have translated into increased complexity regarding the information learned and conveyed through these black-box architectures. Following many researches in neural networks interpretability, we propose in this article a protocol that aims to determine which and where information is located in an ASR acoustic model (AM). To do so, we propose to evaluate AM performance on a determined set of tasks using intermediate representations (here, at different layer levels). Regarding the performance variation and targeted tasks, we can emit hypothesis about which information is enhanced or perturbed at different architecture steps. Experiments are performed on both speaker verification, acoustic environment classification, gender classification, tempo-distortion detection systems and speech sentiment/emotion identification. Analysis showed that neural-based AMs hold heterogeneous information that seems surprisingly uncorrelated with phoneme recognition, such as emotion, sentiment or speaker identity. The low-level hidden layers globally appears useful for the structuring of information while the upper ones would tend to delete useless information for phoneme recognition.
How Important Is Tokenization in French Medical Masked Language Models?
Feb 22, 2024Yanis Labrak, Adrien Bazoge, Beatrice Daille, Mickael Rouvier, Richard Dufour
Subword tokenization has become the prevailing standard in the field of natural language processing (NLP) over recent years, primarily due to the widespread utilization of pre-trained language models. This shift began with Byte-Pair Encoding (BPE) and was later followed by the adoption of SentencePiece and WordPiece. While subword tokenization consistently outperforms character and word-level tokenization, the precise factors contributing to its success remain unclear. Key aspects such as the optimal segmentation granularity for diverse tasks and languages, the influence of data sources on tokenizers, and the role of morphological information in Indo-European languages remain insufficiently explored. This is particularly pertinent for biomedical terminology, characterized by specific rules governing morpheme combinations. Despite the agglutinative nature of biomedical terminology, existing language models do not explicitly incorporate this knowledge, leading to inconsistent tokenization strategies for common terms. In this paper, we seek to delve into the complexities of subword tokenization in French biomedical domain across a variety of NLP tasks and pinpoint areas where further enhancements can be made. We analyze classical tokenization algorithms, including BPE and SentencePiece, and introduce an original tokenization strategy that integrates morpheme-enriched word segmentation into existing tokenization methods.
DrBenchmark: A Large Language Understanding Evaluation Benchmark for French Biomedical Domain
Feb 20, 2024Yanis Labrak, Adrien Bazoge, Oumaima El Khettari, Mickael Rouvier, Pacome Constant dit Beaufils, Natalia Grabar, Beatrice Daille, Solen Quiniou, Emmanuel Morin, Pierre-Antoine Gourraud, Richard Dufour
The biomedical domain has sparked a significant interest in the field of Natural Language Processing (NLP), which has seen substantial advancements with pre-trained language models (PLMs). However, comparing these models has proven challenging due to variations in evaluation protocols across different models. A fair solution is to aggregate diverse downstream tasks into a benchmark, allowing for the assessment of intrinsic PLMs qualities from various perspectives. Although still limited to few languages, this initiative has been undertaken in the biomedical field, notably English and Chinese. This limitation hampers the evaluation of the latest French biomedical models, as they are either assessed on a minimal number of tasks with non-standardized protocols or evaluated using general downstream tasks. To bridge this research gap and account for the unique sensitivities of French, we present the first-ever publicly available French biomedical language understanding benchmark called DrBenchmark. It encompasses 20 diversified tasks, including named-entity recognition, part-of-speech tagging, question-answering, semantic textual similarity, and classification. We evaluate 8 state-of-the-art pre-trained masked language models (MLMs) on general and biomedical-specific data, as well as English specific MLMs to assess their cross-lingual capabilities. Our experiments reveal that no single model excels across all tasks, while generalist models are sometimes still competitive.
BioMistral: A Collection of Open-Source Pretrained Large Language Models for Medical Domains
Feb 15, 2024Yanis Labrak, Adrien Bazoge, Emmanuel Morin, Pierre-Antoine Gourraud, Mickael Rouvier, Richard Dufour
Large Language Models (LLMs) have demonstrated remarkable versatility in recent years, offering potential applications across specialized domains such as healthcare and medicine. Despite the availability of various open-source LLMs tailored for health contexts, adapting general-purpose LLMs to the medical domain presents significant challenges. In this paper, we introduce BioMistral, an open-source LLM tailored for the biomedical domain, utilizing Mistral as its foundation model and further pre-trained on PubMed Central. We conduct a comprehensive evaluation of BioMistral on a benchmark comprising 10 established medical question-answering (QA) tasks in English. We also explore lightweight models obtained through quantization and model merging approaches. Our results demonstrate BioMistral's superior performance compared to existing open-source medical models and its competitive edge against proprietary counterparts. Finally, to address the limited availability of data beyond English and to assess the multilingual generalization of medical LLMs, we automatically translated and evaluated this benchmark into 7 other languages. This marks the first large-scale multilingual evaluation of LLMs in the medical domain. Datasets, multilingual evaluation benchmarks, scripts, and all the models obtained during our experiments are freely released.
Jeffreys divergence-based regularization of neural network output distribution applied to speaker recognition
Dec 28, 2023Pierre-Michel Bousquet, Mickael Rouvier
A new loss function for speaker recognition with deep neural network is proposed, based on Jeffreys Divergence. Adding this divergence to the cross-entropy loss function allows to maximize the target value of the output distribution while smoothing the non-target values. This objective function provides highly discriminative features. Beyond this effect, we propose a theoretical justification of its effectiveness and try to understand how this loss function affects the model, in particular the impact on dataset types (i.e. in-domain or out-of-domain w.r.t the training corpus). Our experiments show that Jeffreys loss consistently outperforms the state-of-the-art for speaker recognition, especially on out-of-domain data, and helps limit false alarms.
SynVox2: Towards a privacy-friendly VoxCeleb2 dataset
Sep 12, 2023Xiaoxiao Miao, Xin Wang, Erica Cooper, Junichi Yamagishi, Nicholas Evans, Massimiliano Todisco, Jean-François Bonastre, Mickael Rouvier

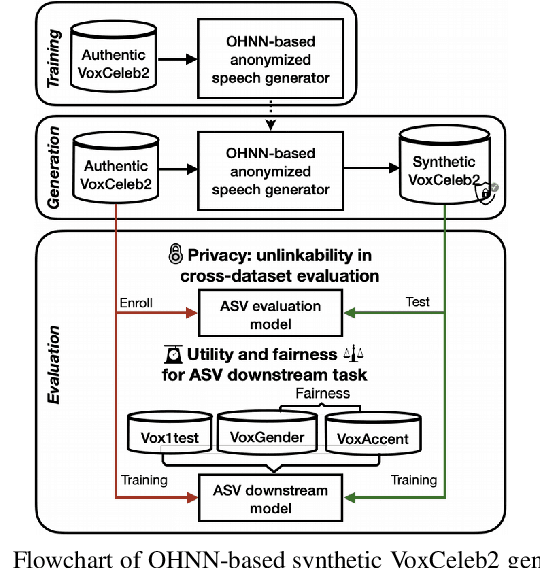
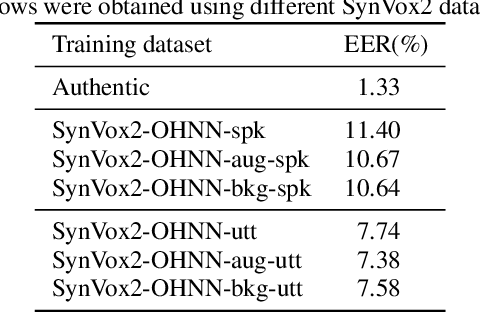
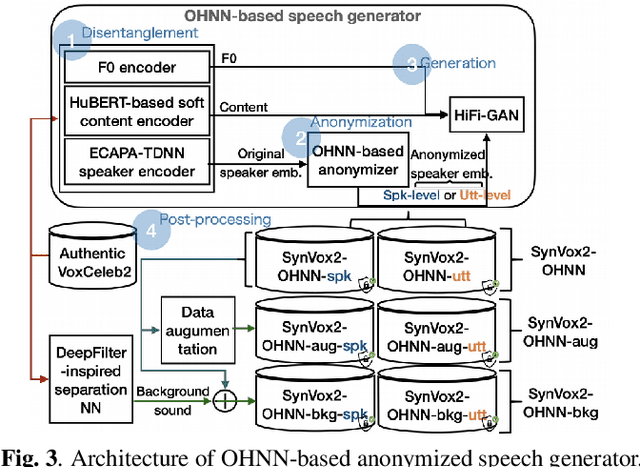
The success of deep learning in speaker recognition relies heavily on the use of large datasets. However, the data-hungry nature of deep learning methods has already being questioned on account the ethical, privacy, and legal concerns that arise when using large-scale datasets of natural speech collected from real human speakers. For example, the widely-used VoxCeleb2 dataset for speaker recognition is no longer accessible from the official website. To mitigate these concerns, this work presents an initiative to generate a privacy-friendly synthetic VoxCeleb2 dataset that ensures the quality of the generated speech in terms of privacy, utility, and fairness. We also discuss the challenges of using synthetic data for the downstream task of speaker verification.
LeBenchmark 2.0: a Standardized, Replicable and Enhanced Framework for Self-supervised Representations of French Speech
Sep 11, 2023Titouan Parcollet, Ha Nguyen, Solene Evain, Marcely Zanon Boito, Adrien Pupier, Salima Mdhaffar, Hang Le, Sina Alisamir, Natalia Tomashenko, Marco Dinarelli, Shucong Zhang, Alexandre Allauzen, Maximin Coavoux, Yannick Esteve, Mickael Rouvier, Jerome Goulian, Benjamin Lecouteux, Francois Portet, Solange Rossato, Fabien Ringeval, Didier Schwab, Laurent Besacier

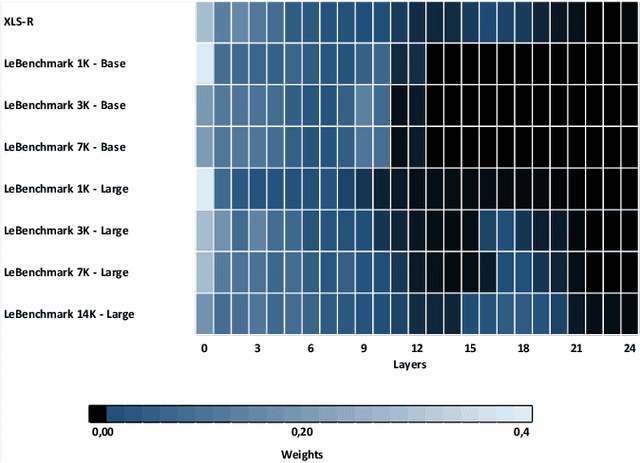
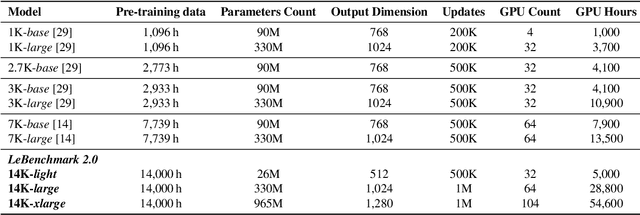
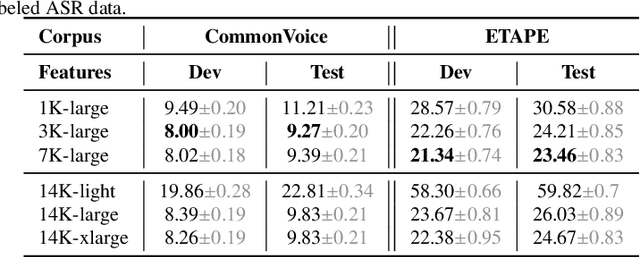
Self-supervised learning (SSL) is at the origin of unprecedented improvements in many different domains including computer vision and natural language processing. Speech processing drastically benefitted from SSL as most of the current domain-related tasks are now being approached with pre-trained models. This work introduces LeBenchmark 2.0 an open-source framework for assessing and building SSL-equipped French speech technologies. It includes documented, large-scale and heterogeneous corpora with up to 14,000 hours of heterogeneous speech, ten pre-trained SSL wav2vec 2.0 models containing from 26 million to one billion learnable parameters shared with the community, and an evaluation protocol made of six downstream tasks to complement existing benchmarks. LeBenchmark 2.0 also presents unique perspectives on pre-trained SSL models for speech with the investigation of frozen versus fine-tuned downstream models, task-agnostic versus task-specific pre-trained models as well as a discussion on the carbon footprint of large-scale model training.
A Zero-shot and Few-shot Study of Instruction-Finetuned Large Language Models Applied to Clinical and Biomedical Tasks
Jul 22, 2023Yanis Labrak, Mickael Rouvier, Richard Dufour
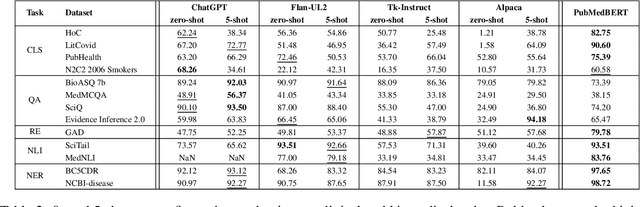
We evaluate four state-of-the-art instruction-tuned large language models (LLMs) -- ChatGPT, Flan-T5 UL2, Tk-Instruct, and Alpaca -- on a set of 13 real-world clinical and biomedical natural language processing (NLP) tasks in English, such as named-entity recognition (NER), question-answering (QA), relation extraction (RE), etc. Our overall results demonstrate that the evaluated LLMs begin to approach performance of state-of-the-art models in zero- and few-shot scenarios for most tasks, and particularly well for the QA task, even though they have never seen examples from these tasks before. However, we observed that the classification and RE tasks perform below what can be achieved with a specifically trained model for the medical field, such as PubMedBERT. Finally, we noted that no LLM outperforms all the others on all the studied tasks, with some models being better suited for certain tasks than others.
FrenchMedMCQA: A French Multiple-Choice Question Answering Dataset for Medical domain
Apr 09, 2023Yanis Labrak, Adrien Bazoge, Richard Dufour, Mickael Rouvier, Emmanuel Morin, Béatrice Daille, Pierre-Antoine Gourraud


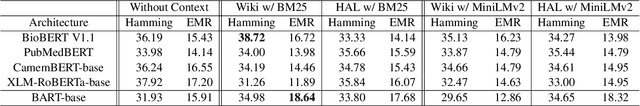
This paper introduces FrenchMedMCQA, the first publicly available Multiple-Choice Question Answering (MCQA) dataset in French for medical domain. It is composed of 3,105 questions taken from real exams of the French medical specialization diploma in pharmacy, mixing single and multiple answers. Each instance of the dataset contains an identifier, a question, five possible answers and their manual correction(s). We also propose first baseline models to automatically process this MCQA task in order to report on the current performances and to highlight the difficulty of the task. A detailed analysis of the results showed that it is necessary to have representations adapted to the medical domain or to the MCQA task: in our case, English specialized models yielded better results than generic French ones, even though FrenchMedMCQA is in French. Corpus, models and tools are available online.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge